Being a biology major, most my time is spent in lab. In addition to lectures, almost all sciences courses require a four-hour long lab once a week (though sometimes we don’t use the whole four hours). And being a Senior, I have taken a fair amount of science courses. Looking back at the courses I took, I am astounded by the variety of labs we’ve done. So I wanted to make a list of all the memorable labs/experiments for each science course I have taken/am taking (some with pictures!).
(Since this list is pretty long, I have divided them up.)
Most memorable experiment done in lab (Sophomore Year)
BIO 111 – Biofilm*
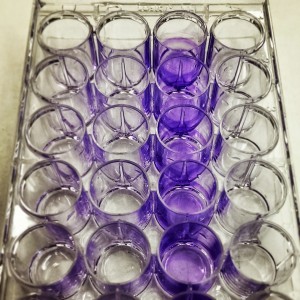
We tested the effectiveness of four different treatment (enzyme amylase, cranberry juice, honey, kanamycin (antibiotic) on biofilm formation of the bacteria Pseudomonas putida. To test this, we used crystal violet dye which stains biofilm a vibrant purple color, so the amount of dye correlates to the amount of biofilm formed.
*biofilm is a biological layer made up of a group of microorganisms that stick together and adhere to a surface
BIO 112 – Bird Flight



We measured the amount of lift that was exerted by different bird species wings. This lab was memorable because of this crazy “contraption” (aka a wind tunnel) we used to measure lift. It was such an unusual sight see. There were these just giant, coloured, plastic tubes set on the table tops. Inside these wind tunnels we would then clip in the wings, which was connected to a balance to determine the amount of lift.
BIO 211 – Species Richness



This was one of the few labs that did not require actually being inside of a lab, and was more of like a “field trip.” In this lab, we looked at species richness* in intertidal zones. To do this, we took a little trip to an area close to Fox Island. Under the Bridgeway there was just a plethora of aquatic organisms: anemones, crabs, mussels, and star fishes (just to name a few).
* the amount of different species
CHEM 120 – Synthesis and Recrystallization of Aquapentaamminecobalt(III) nitrate
In this lab, we created cobalt(III) complexes by oxidizing cobalt(II) salts with hydrogen peroxide, and using ammonium hydroxide and ammonium nitrate for our ammonia source. We then used recrystallization to isolate our desired cobalt(III) product, which involved increasing pH of the solution. This was my first occurrence with recrystallization, so it was interesting to be able to see crystals forming as it cooled in the ice bath.
Click here for part II.
*Excuse me for the poor image quality, they were taken when the Iphone 5c was the new “it” phone